Reclaiming Childhood
Duke Children’s Gene Therapy is Giving Kids with Sickle Cell Disease a Future Free from Pain.
Until recently, a diagnosis of sickle cell disease too often meant a life defined by pain, frequent hospital stays, and uncertainty—but Duke Children’s is rewriting that story.
Sickle cell disease is an inherited blood disorder. Though diagnosed at birth, the disease's effects—caused by rigid, sickle-shaped cells blocking blood flow—build over time, leading to chronic anemia, organ damage and unpredictable pain episodes known as vaso-occlusive crises. For pediatric patients and their families, a sickle cell diagnosis is the first step in a lifelong journey, turning caregivers into fierce advocates who stand ready for whatever comes.
At Duke Children’s, however, a new era of treatment has arrived, offering the promise of lasting relief and renewed possibility. As one of the few centers in the nation—and the first in the Southeast—to offer the newly FDA-approved gene therapy, Lyfgenia, Duke Health is bringing hope to patients. This revolutionary treatment uses the patient’s own stem cells to eliminate the disease's acute effects and unlock a future of a healthy, unrestricted life.
The therapy is a major advancement compared to traditional bone marrow transplantation. While curative, bone marrow transplantation often utilizes a perfectly matched sibling donor—something only a small percentage of patients have. For all others, Duke Children’s use of gene therapy now offers new hope.
A CHILDHOOD INTERRUPTED BY CHRONIC PAIN
For Aaron Scott of Fayetteville, North Carolina, the need for a solution was urgent and undeniable. While the impact of sickle cell disease on Aaron had been relatively mild in early childhood, his condition escalated quickly in adolescence. His physician, Kris Mahadeo, MD, MPH, who leads Duke Children's Pediatric Transplant and Cellular Therapy (PTCT) program, notes that Aaron was a classic example of the disease's unpredictable nature.
“Unfortunately, as Aaron matured, the impact of sickle cell disease grew progressively severe and his pain crises intensified,” said Mahadeo.
Aaron and his grandmother, Stephanie Scott-Miller, together endured years of managing the disease with standard therapies, only to watch the pain take over. The crises became so frequent and severe that Aaron was unable to attend school in-person.
“In 18 months, Aaron was hospitalized more than a dozen times,” Scott-Miller recalls. “Despite his best efforts, the disease kept him from a normal high school experience filled with sports, friends, and fun.”
The unpredictable pain could strike anywhere—his back, ribs, shoulders, or legs. Explaining the agony, Aaron described it as the feeling of broken glass traveling through his body. The episodes were not just physically agonizing; they were mentally draining. “I just didn’t like life back then,” Aaron shared.
Sickle cell disease relentlessly steals childhood and reduces life expectancy—a reality Duke Children’s is determined to change.
"The Duke team managed the complex financial process—but their dedication extended far beyond that," Scott-Miller says. "After years of struggling at our local hospital, Duke Children’s looked at Aaron as a person with a bright future, not as a patient seeking medication. They provided him with a complete network of champions—social workers, therapists, and even a tutor so he could finish high school."
Stephanie Scott-Miller
GENE THERAPY: A REVOLUTION FROM WITHIN
When traditional options failed, Scott-Miller began researching alternatives and discovered Duke Children’s sickle cell disease research. With help from the Duke Pediatric PTCT and hematology teams, Aaron and his grandmother waited for the FDA’s approval of the gene therapy, Lyfgenia. When its use was approved in December 2023 for patients 12 and older, Aaron became an immediate candidate and Duke Children’s was ideally positioned to support him.
The concept behind gene therapy is revolutionary: help the body heal itself. Mahadeo explains the process simply, “Through a series of intricate steps, we harvest the patient’s stem cells. These are then transformed to produce normal, non-sickling hemoglobin. Using the recipient's own modified cells avoids the serious risk of graft-versus-host disease, a complication that can occur when transplanted cells from a donor attack a recipient’s healthy tissues as may occur with traditional bone marrow transplant.”
GUIDING EVERY STEP: THE POWER OF COMPASSIONATE COORDINATION
The decision to pursue gene therapy marks only the first step in a months-long process. Walking side-by-side with families throughout, offering logistical and emotional support, is the dedicated PTCT team, which included Aaron’s nurse coordinator, Emily Dodd, BSN, RN.
Treatment management is a Herculean undertaking, especially given the significant financial hurdle. The gene therapy products alone come at an extremely high cost, exceeding $2 million. However, the institutional resolve at Duke Children's means this burden doesn't fall on the family. Scott-Miller credits Aaron's care team for their fierce advocacy, working with state lawmakers and Medicaid to gain approval for the complex funding.
"The Duke team managed the complex financial process—but their dedication extended far beyond that," Scott-Miller says. "After years of struggling at our local hospital, Duke Children’s looked at Aaron as a person with a bright future, not as a patient seeking medication. They provided him with a complete network of champions—social workers, therapists, and even a tutor so he could finish high school."
A SEASON OF HOPE AND HEALING
The first milestone was stem cell collection. Over five days in the hospital, Aaron underwent apheresis, a procedure that collected his healthy, blood-forming cells for modification. The cells were then sent for processing, where scientists reengineered them to produce normal hemoglobin. For Aaron and his family, the next 10 weeks—while the cells were transformed—were filled with hope and anticipation.
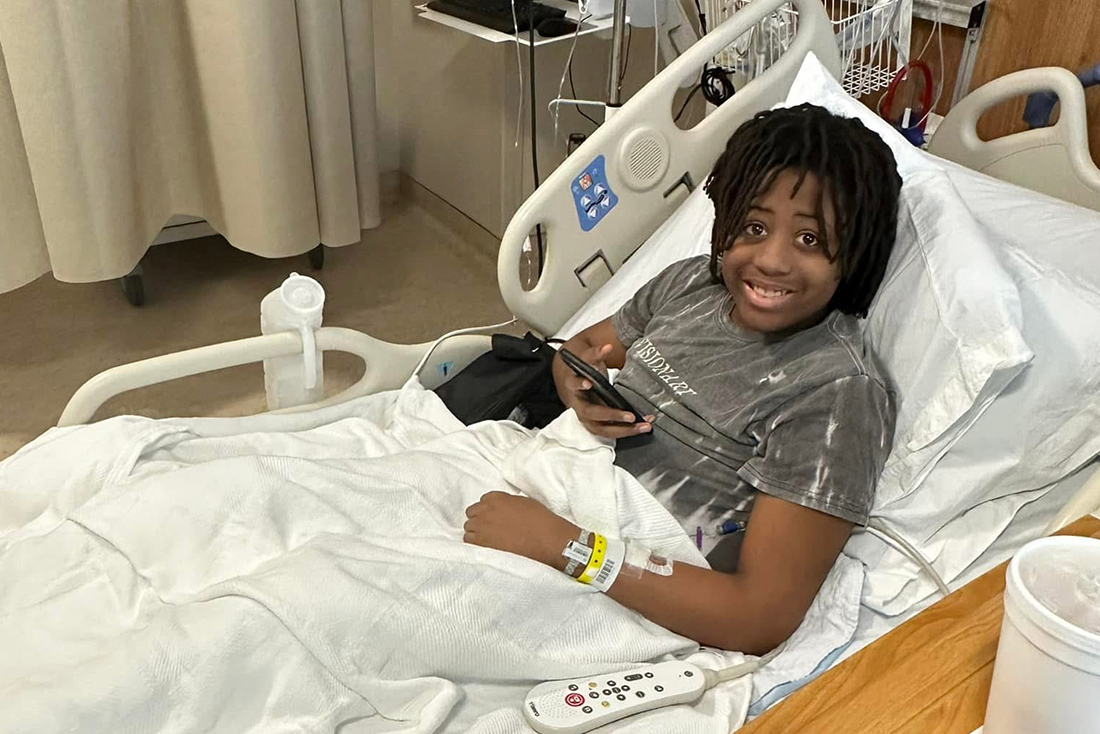
The most intense phase began in January 2025. Aaron was admitted for the transplant, a process that required four days of chemotherapy to safely clear out his existing bone marrow. Then came the infusion: the momentous day the gene-modified cells were administered. The subsequent wait for engraftment—the moment the new cells successfully take root—was filled with anxiety, but Aaron’s recovery was remarkably swift, leading to his discharge just 30 days post infusion. Every step of this journey underscores Aaron’s courage and the compassionate coordination provided by his care team.
A NEW LIFE: FROM PAIN CRISIS TO THE GYM
Since receiving his gene therapy, 18-year-old Aaron, has not experienced a single pain crisis.
“He hasn’t had a single pain crisis since receiving the gene therapy—and we expect more than 90% of patients to share Aaron’s experience,” Mahadeo said confidently. “This treatment is truly transformative, the change in quality of life for the patient is immediate and profound.”
Dodd keeps a close eye on her patients’ progress. “Seeing patients reclaim their lives is deeply rewarding,” said Dodd. “We’re helping them achieve what once seemed impossible.”
Aaron himself is embracing his newfound freedom: “I play basketball and work out now,” he said. “Even being outside in the cold—something I used to fear—is no longer a problem.”
DUKE’S COMMITMENT: SUPPORT AND THE FUTURE OF CARE
Aaron’s experience showcases more than cutting-edge therapies, but also Duke Children’s unique, comprehensive approach to patient care. As Mahadeo noted, ensuring patients and their families can access and complete this arduous treatment requires a massive cross-institutional effort.
For families traveling from afar—like Aaron’s four-hour round trip—the PTCT Family Support Program is essential. Patients must remain close to Duke for 60 to 100 days post-infusion. This provision lets families focus on healing, not logistics. As a nurse coordinator, Dodd emphasizes this institutional goal: “By helping to remove logistical barriers that often accompany intense treatment, we help families maintain a sense of stability and ensure the child remains comfortable.”
Looking ahead, Duke Children’s is leading trials for younger patients to deliver therapy earlier—before lasting damage occurs.
The pressing need for resources is constant. Funding is critical, not just for the high cost of therapies, but for research into optimizing care and the well-bring of families whose lives are upended during the treatment period. Philanthropy is vital to expanding Duke's ability to offer this curative treatment and accelerate the timeline for every eligible child.
Aaron’s transformation offers a powerful glimpse into the future of sickle cell care—and hope for countless others still waiting for relief. His message to them mirrors his care team’s unwavering belief: “Don’t give up.”
UNLOCKING A LIFE WITHOUT LIMITS
Lyfgenia, the FDA-approved gene therapy product used to cure sickle cell disease, comes with a high price, costing over $2 million per patient. Unfortunately, this figure only covers over the therapeutic product itself and does not include the extensive hospitalization, other essential medications or the comprehensive family care provided throughout the treatment and recovery process. Philanthropy is essential to removing financial barriers and enabling Duke Children’s to expand its groundbreaking work and rewrite the story for other children like Aaron.
At Duke Children’s, the goal remains: ensure every child with sickle cell disease has the chance to live a pain-free, fully realized life.
YOUR GIFT CREATES A BRIGHTER FUTURE FOR CHILDREN IN NEED
We appreciate gifts of any size, making sure that your generosity is used to advance medical care and provide a better tomorrow for our children. You can choose a dollar amount to give now, or continue exploring more ways that you can support Duke Children’s.